A list of 43 important hydroponic fertilizers
First it is important to understand which nutrients are included in the various fertilizers and in what amounts. It is important to distinguish between macro and micro elements. The macro elements are taken up in large quantities and the micro elements in smaller quantities. Although the micro elements are taken up by smaller amounts, it does not mean that they are less important. Many growers neglect micro element management with the result that plants are less resistant to disease and yields suffer. The macro elements are: Nitrogen (N), Phosphorus (P), Potassium (K), Calcium (Ca), Magnesium (Mg), Sulphur (S), Oxygen (O), Carbon (C) and Hydrogen (H). The micro elements are: Iron (Fe), Boron (B), Manganese (Mn), Zinc (Zn), Copper (Cu) and Molybdenum (Mo).
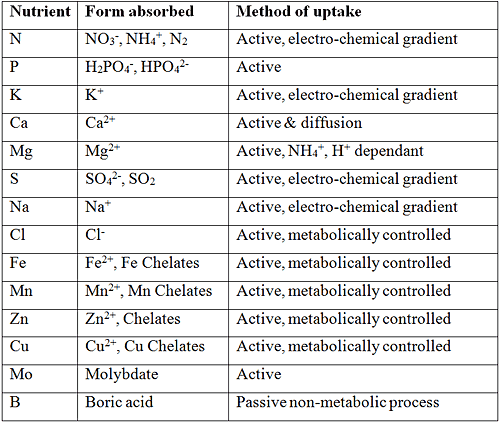
The form in which various nutrients are absorbed by plants is important and helps to understand which fertilizers to use. For instance, nitrite (NO2-) is extremely toxic to plants and should never be applied as a fertilizer. Plants cannot absorb Fe3+ since it precipitates. Fe is absorbed as Fe2+ with the help of a chelating agent. The form in which the macro and micro elements are absorbed by plant roots are listed in the table below. The method of uptake is also important and determined quite often the interaction, be it synergistic or antagonistic between various elements.
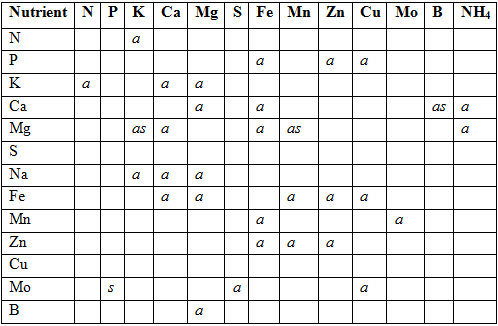
By applying to much of, for instance ammonium, will depress the uptake of both magnesium and calcium (antagonistic effect), on the other hand, the toxic effect of B can be reduced by increasing the Ca concentration of the nutrient solution (synergistic effect).
The range of fertilizers available to the grower is much more than conventional granular fertilizers. Some of these fertilizers are used by fertilizer companies to make up complex mixes or prescription mixes. A complete nutrient formulation can be made from the fertilizers listed below:
- Potassium nitrate (KNO3) 62.90% K
- Calcium nitrate (CaNO3.4H2O) 24.39% Ca
- Sodium nitrate (NaNO3) 72.94% N
- Ammonium sulphate ((NH4)2SO4) 21.33% N
- Ammonium nitrate (stabilized) (NH4NO3 ) 35% N
- Urea (CO(NH2)2 ) 46.67% N
- Am. dihydrogen phosphate (MAP) (NH4H2PO4 ) 12.17% N
- Di-Am. Dihydrogen phosphate (DAP) ((NH4)2HPO4 ) 24.24% N
- Potassium dihydrogen phosphate (KH2PO4) 22.79% P
- Monocalcium phosphate (CaH4(PO4)2.H2O) 24.6% P
- Phosphoric acid (H3PO4) 31.63% P
- Potassium sulphate (K2SO4) 44.83% K
- Potassium chloride (KCl) 50% K
- Calcium sulphate (CaSO4.2H2O) 23.26% Ca
- Magnesium sulphate (MgSO4.7H2O) 9.75% Mg
- Magnesium nitrate (Mg(NO3)2.6H2O ) 9.38% Mg
- Na-Fe EDTA (Chelate) 4.1043% Fe
- Na-Fe HEDTA (Chelate) 4.1038% Fe
- Na-Fe EDDHA (Chelate) 6% Fe
- Na-Fe DTPA (Chelate) 10% Fe
- Ferrous sulphate (green vitriol) (FeSO4.7H2O) 19% Fe
- Ferric sulphate (Fe2(SO4)3.4H2O) 23% Fe
- Ferrous oxide (FeO) 77% Fe
- Ferric oxide (Fe2O3) 69% Fe
- Ferrous ammonium phosphate (Fe(NH4)PO4.H2O) 29% Fe
- Ferrous ammonium sulphate ((NH4)2SO4.FeSO4.6H2O) 14% Fe
- Iron ammonium polyphosphate (Fe(NH4)HP2O7) 22% Fe
- Boric acid (H3BO3) 17.74% B
- Solubor (Na2B4O7.5H2O + Na2B10O16.10H2O) 20.5% B
- Sodium tetra borate (Na2B4O7.10H2O) 2.83% B
- Copper sulphate (CuSO4.5H2O) 25.6% Cu
- Copper sulphate monohydrate (CuSO4.H2O) 35% Cu
- Cu Chelate (Na2CuEDTA) 13% Cu
- Cu Chelate (NaCuHEDTA) 9% Cu
- Ammonium molybdate ((NH4)6.Mo7O24.4H2O) 54.37% Mo
- Sodium molybdate (Na2MoO4.2H2O) 39% Mo
- Zinc sulphate (ZnSO4.7H2O) 22.65% Zn
- Zinc sulphate monohydrate (ZnSO4.H2O) 35% Zn
- Zn Chelate (Na2ZnEDTA) 14% Zn
- Zn Chelate (NaZnNTA) 13% Zn
- Zn Chelate (NaZnHEDTA) 9% Zn
- Manganese sulphate (MnSO4.4H2O) 24.63% Mn
- Mn Chelate (MnEDTA) 12% Mn
In comparison with conventional granular fertilizers that are applied directly to the soil, hydroponic fertilizers are water soluble and must be dissolved in water before they are applied to the fertigation system. Thus is extremely important. If the fertilizers are not dissolved first, the correct balance and ratio will not reach the plants since some of the nutrients are still insoluble and not available to the plant. The mixing procedure is extremely important.
Share This Story, Choose Your Platform!
Cotton aphid – Aphis gossypii
Cotton Aphid, or Aphis gossypii, is one of the more common insect pests found in almost all vegetable crops and [...]
Greenhouse white fly
Description Greenhouse white fly, also known as Trialeurodes vaporariorum is a sap feeder. The easiest way to find white flies [...]